 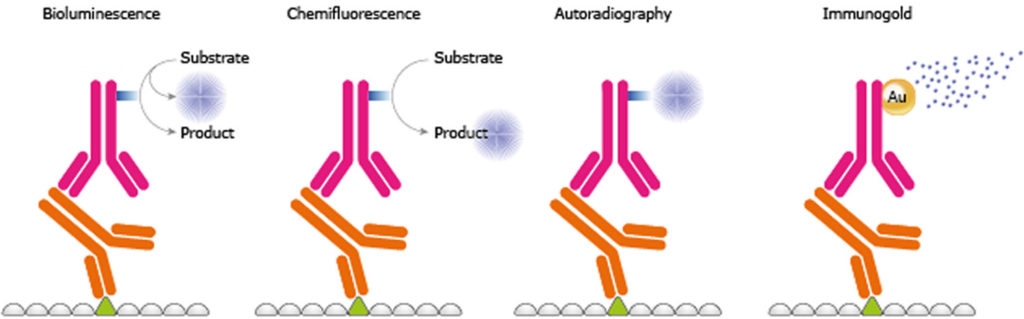
recently recently touched this topic in their editorial in Molecular & Cellular Proteomics and advocated the use of MRM proteomics. Considering the latter, it is important to compare the advantages and disadvantages of both these methods. Therefore, it is a challenge for biologists to see beyond western-blotting, and accept and adopt MRM proteomics in routine protein quantification work. On the other hand, western-blotting (or protein immunoblotting) is a conventional protein quantification method which is a standard technique in almost every biology laboratory. Because of its increasing popularity, Nature Methods declared targeted proteomics by mass spectrometry (MS) as Method of the Year for 2012. For instance, successful completion of human genome project and the ongoing human proteome project enabled us to select surrogate peptides for MRM quantification of nearly every human protein. This application of MS based targeted proteomics become wider with the availability of protein databases and in silico methods for surrogate peptide selection in the last five years. 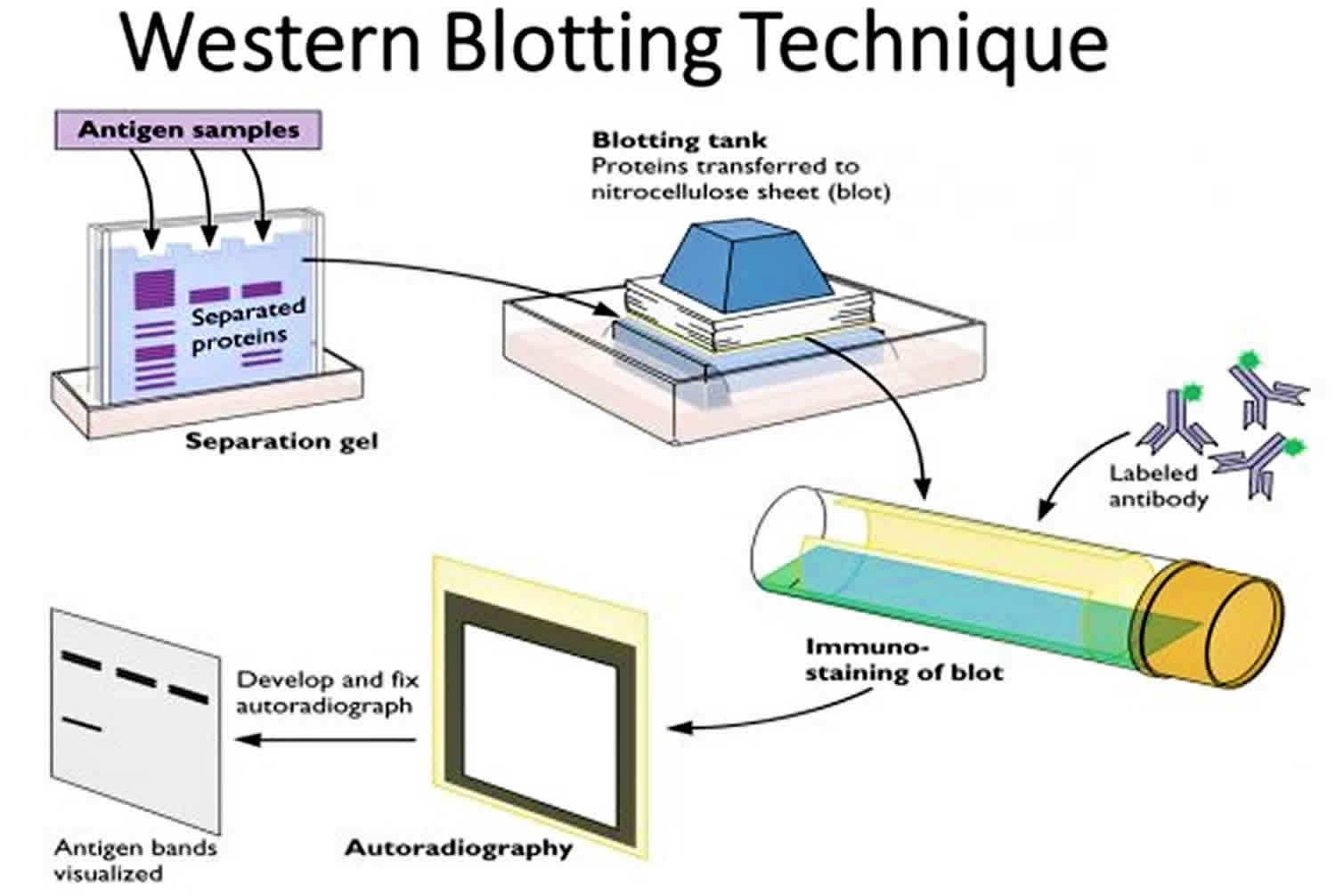
The selectivity is achieved by the MRM characteristic of triple quandrupole MS instrument in which unique daughter ion generated from fragmentation of a parent ion (i.e, ionized analyte peptide) is monitored. MS based protein quantification, commonly referred as multiple or selective reaction monitoring (MRM or SRM) proteomics, relies on selective quantification of surrogate peptide(s) in a digested protein sample. Sensitivity and selectivity of liquid chromatographycoupled with tandem mass spectrometry (LC-MS/MS) for quantification of small molecules encouraged biologists to utilize this platform for protein quantification using peptide as a surrogate. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
